|
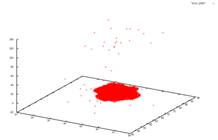
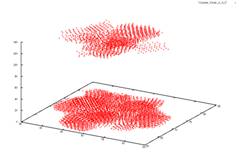
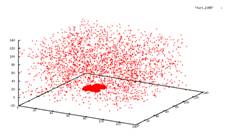
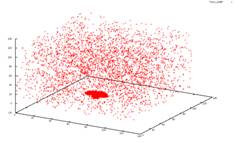
Sublimation in 3D I studied crystal sublimation under the influence of the Asakura-Oosawa potential. The AO potential is a short range interaction, and the specific systems I was dealing with have only a gas-crystal phase transition. We wished to take the data from the simulation and see if we could deduce a mechanism for the sublimation of the system. Possible theories would be that the sublimated particles must be on the surface of the crystal, or on an edge. Perhaps even, all particles in the crystal would have an equal chance at sublimating. However none of these possibilities were supported by the data. In a 2006 paper by Savage et al., it was suggested that in two dimensions crystals form a transient liquid coating, from which the sublimated particles emerge. We are currently studying the implications of this to see if this is a possible mechanism for sublimation in three dimensions. AO systems are useful in the study of globular proteins. These proteins, like AO colloids are often hard spheres. Phase transitions in these proteins then are caused by changes in the entropy of the system. This system shown below has a ratio of the polymer radius and colloid radius of 0.1. The cluster has a size is 5688 particles. The original crystal was formed at 4kT. The system is then heated up to 3kT to see the sublimation. From left to right you see the crystal at times t=0, 50, 5000 and 10000. We employ periodic boundary conditions, so at t=0 it looks like two crystals, but really the crystal is just at the very bottom of the box. The other images do not have this issue because they are at intermediate steps during the run. If you look closely, you can see that the three other graphs go into the negative range.
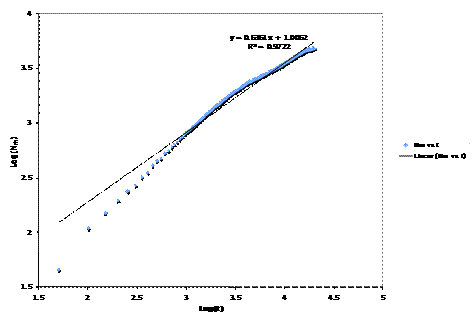
We find that the sublimation can be described approximately by Nm~t2/3 As can be seen in the following graph
This is only one particular system, but all the systems seemed to follow this approximate scaling law.
|
||||
|
|