Optical Fiber Based Systems for the Spectroscopy of Nanoparticles
By: Jeremy Stone
Supervisor:
Dr. Brian Washburn and Dr. Bret Flanders
Kansas
State University
Physics REU 2011
Kansas State University Physics Department REU Program
This program is funded by the National Science Foundation through grant number PHY-0851599.
Welcome to my webpage. This page summarizes my experience doing research in the James R. Macdonald Lab working under Dr. Brian Washburn. I am working with carbon nanotubes and trying different methods to get light to interact with them.
Summary: My goal is to find new ways of doing spectroscopy on small amounts of nanoparticles. The types of nanoparticles I am working with are carbon nanotubes, but hopefully my methods will work with other types of nanoparticles as well. The first half of my project I tried different methods for getting carbon nanotubes into hollow core photonic band gap fibers. This is useful because it will give the light a long length in which to interact with the nanotubes. The second half I spent my time setting up and doing spectral interferometry on single mode fibers. A taper was made in the middle of a section of the fiber and nanotubes were deposited on the outside of the fiber over the tapered area. A pulse of light was sent through and we could see if there was any difference in the output spectrum of the laser.
Project Goals: My first goal for the summer was to find out if it was possible to deposit carbon nanotubes inside of hollow core photonic band gap fibers. If I were successful in doing that, I would then do spectral measurements on the fiber to see if there was any interaction between the laser and nanotubes. My other goal was to build a spectral interferometer in which one area of the fiber was tapered and the nanotubes were deposited on this area. The evanescent waves from the laser passing through a tapered section of the fiber could interact with the nanotubes this way as well.
Research Strategy: My main strategy for depositing the nanotubes inside of the fiber was mainly trial and error. I had a vacuum pump to try to suck a solution of carbon nanotubes and a florescent dye out of a syringe. I would try to do it one way and then examine the end of the fiber and the side of it to see if I could see any evidence of the solution inside. A laser pointer was also used to fluoresce the dye to help see if the solution was inserted. Me and my supervisor would then try to think of why it might not be working and try to think of a solution to the problem.
Research
Progress: I
spent a lot of my time at first setting up my design on a breadboard. I set up a stand for my laser point and two
mirrors that direct the laser into a lens.
The lens focuses the laser onto one end of my fiber that is being held
inside of a vacuum chamber. The vacuum
chamber has a positive pressure and a negative pressure line on it to move the
solution back and forth inside the fiber. The other end of the fiber is
inserted into a syringe that contains my solution. A microscope is set up near
the end of the fiber where the nanotubes are being deposited. The vacuum chamber and lens are on a linear
stage so that I can move the fiber back and forth in front of the microscope to
see different areas. I could also shift
around a few of my optical elements to look at the end of the fiber to see the
cross section.

Figure 1: Photograph of my set up
described above.
I tried making different ratios of carbon nanotubes solution to fluorescent dye to see if there was an optimal ratio. I sonicated each of them for at least an hour before trying to use them, but during the sonication process an unknown chemical reaction would take place and turn the once transparent solution into an opaque solution. I tried using it anyways to see if it would still work.

Figure 2: Side view of my fiber with CNT and fluorescein inside.
The white dots are either laser light scattering off the solution/air
interface or the dye emitting light.
Since the light in photonic band gap fibers travels down the central core of the fiber, I wanted to taper the fiber to close off all the holes except the central hole. This would cause my solution to only be deposited in the area were the light is.


Figure 3: Photo of the photonic band
gap fiber (Blaze Photonics “Hollow Core Photonic Band Gap Fiber” Issue 16/2/4)
and of my fiber with the laser on without nanotubes.
The problem that I had after depositing the nanotubes was that I wasn’t able to see any light coming from the core. The nanotubes seemed to be absorbing all the light that entered the fiber. This is when me and my supervisor decided to try a different way of doing spectroscopy on the nanotubes.
I then switched to making a spectral interferometer and depositing nanotubes onto a tapered section of a single mode fiber. I could then look at a before and after at the same time delays with and without nanotubes to see if any modulations or time shifts occurred.
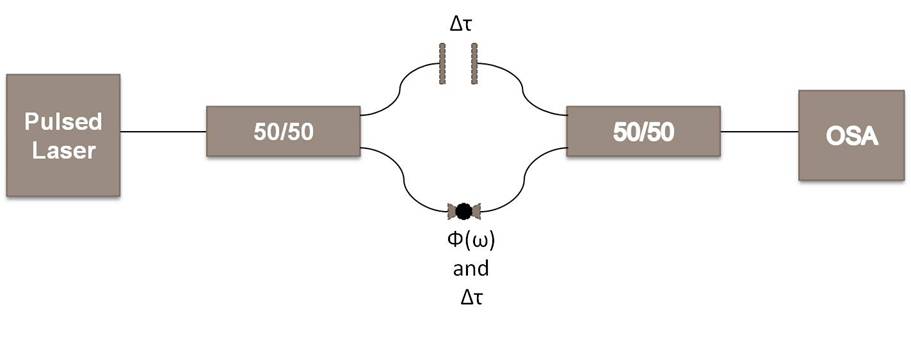
Figure 4: Diagram of my spectral
interferometer.
Once each arm in the interferometer were close to the same length I could start taking some data. I took measurements on the optical spectrum analyzer at different time delays.
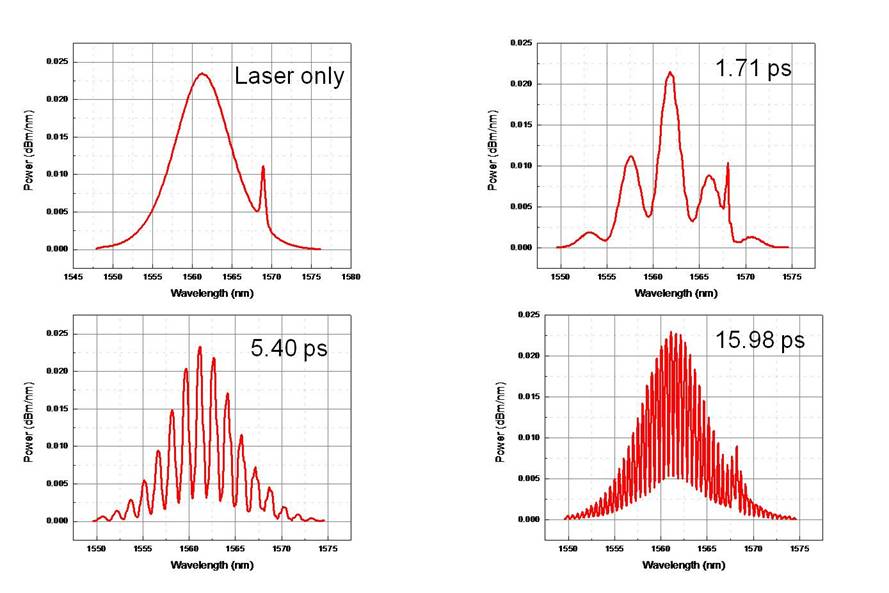
Figure 5: Different fringe patterns made at different
time delays.
I wasn’t sure if the nanotubes actually got deposited on the fiber or not so I tried taking new measurements with a solution of nanotubes that was about 3.5 times as concentrated. With the more concentrated solution, modulation in the fringes could be seen.
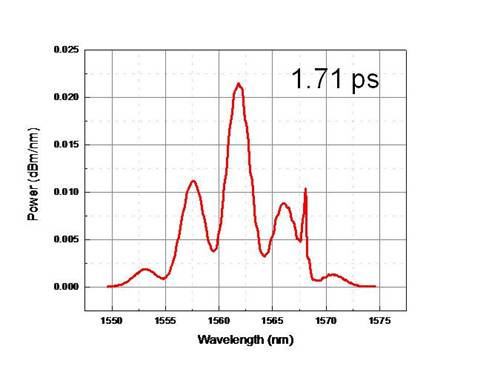
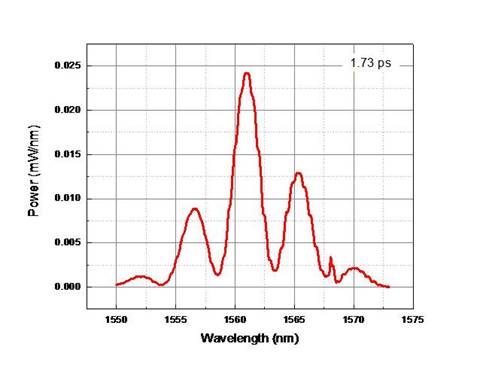
Figure 6: Comparison of OSA before and
after depositing the 3.5x concentrated solution. Modulations are hard to see, but
are present in the 2nd graph.
Final Presentation: Click here to download my final presentation
About Me: I attend school at Northern Arizona University. I am going to be a fifth year student, graduating next year. I enjoy hiking, camping, snowboarding, and pretty much anything else outside.
Check out these useful sites: